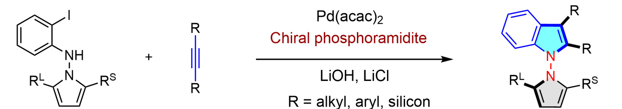
Pd-catalyzed asymmetric Larock reaction for the atroposelective synthesis of N─N chiral indoles
Atropisomeric indoles defined by a N─N axis are an important class of heterocycles in synthetic and medicinal chemistry and material sciences. However, they remain heavily underexplored due to limited synthetic methods and challenging stereocontrol over the short N─N bonds. Here, we report highly atroposelective access to N─N axially chiral indoles via the asymmetric Larock reaction. This protocol leveraged the powerful role of chiral phosphoramidite ligand to attenuate the common ligand dissociation in the original Larock reaction, forming N─N chiral indoles with excellent functional group tolerance and high enantioselectivity via palladium-catalyzed intermolecular annulation between readily available o- iodoaniline and alkynes. The multifunctionality in the prepared chiral indoles allowed diverse post-coupling synthetic transformations, affording a broad array of functionalized chiral indoles. Experimental and computational studies have been conducted to explore the re-action mechanism, elucidating the enantio-determining and rate- limiting steps.
(Sci .Adv. 2024. DOI: 10.1126/sciadv.ado4489.)
Congratulations to Jinlei Wang. Special thanks also go to our collaborators.