Copper-Catalyzed Chemoselective (Amino)fluorosulfonylation of Hydrocarbons via Intramolecular Fluorine-Atom Transfer
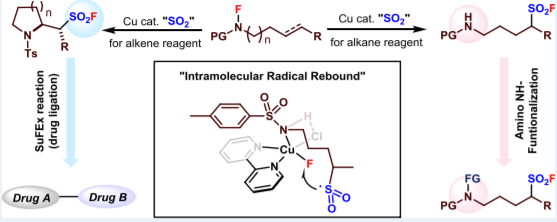
Sulfonyl fluorides have found increasing applications as functional molecules in chemistry and biology. We herein report a copper-catalyzed atom-economical access to two categories of sulfonyl fluorides through a radical relay strategy in the presence of an SO2 surrogate. The aliphatic C(sp3 )−H bond in N-fluoro-N-alkyl sulfonamides reacted via a 1,5-hydrogen atom transfer (HAT) process, affording alkanesulfonyl fluorides with a proximal amino group. On the other hand, utilizing substrates containing a proper C-C double bond resulted in intramolecular olefin aminofluorosulfonylation, allowing the synthesis of fluorosulfonyl-functionalized pyrrolidines and piperidines via atom-transfer radical addition (ATRA). Both reaction systems proceeded under mild conditions, requiring no additional fluorine source. Experimental and computational studies suggest that S−F coupling is likely achieved through an intramolecular radical-rebound pathway. By taking advantage of the SuFEx chemistry and multifunctionality of the products, the method is applicable to the late-stage modification of bioactive compounds, drug ligation chemistry, and organic synthesis.(Cooperated with the team of Shandong University)
(ACS Catal. 2024, 14, 4318−4328)
Congratulations to Shuting Qu. Special thanks also go to our collaborators.